~~~ Probiotics deliver Cellular Bio-Availability. Vitamins Protocol provide Healing Nutrients. Synergy for Cure. Heal Chronic Conditions. ~~~
Probiotic stability is a quantum process ...
Probiotic stability is about keeping the eficacy of probiotics over effective storage time.
“Probiotics are live microorganisms (e.g., bacteria) that are either the same as or similar to microorganisms found naturally in the human body and may be beneficial to health. Also referred to as "good bacteria" or "helpful bacteria," probiotics are available to consumers in oral products such as dietary supplements and yogurts, as well as other products such as suppositories and creams. The body, especially the lower gastrointestinal tract (the gut), contains a complex and diverse community of bacteria. (In the body of a healthy adult, cells of microorganisms are estimated to outnumber human cells by a factor of ten to one.) Although we tend to think of bacteria as harmful "germs," many bacteria actually help the body function properly. Most probiotics are bacteria similar to the beneficial bacteria found naturally in the human gut.”1
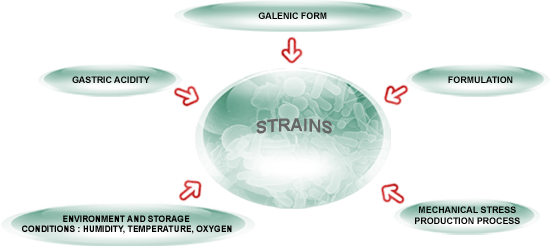
Probiotics stability are measured in colony forming units (CFU). CFUs are generally measured in the millions or billions per serving. Probiotics are most commonly beneficial bacteria, but can also be friendly fungal or other organisms, that are typically freeze dried to stabilize them in an inert state during storage and production. Then their continued stability and viability, as measured by CFU counts when cultured, is dependent on limiting their exposure to stimulating environmental conditions such as warmth and moisture. Besides refrigeration, this protection can be done by packaging in glass, as well as by adding freshness packets that help to absorb and reduce moisture in the package.
Temperature plays a role in probiotic stability. Colder air holds less moisture and is not in the ideal temperature range for the bacteria to commonly grow and thrive, thus inhibiting reactivation of the dormant organisms by depriving them of the warmth and moisture that represent their ideal growing conditions. High heat can also degrade the viability of these organisms.
Even under ideal storage conditions, the number of colony forming units will slowly decline as months go on. For example, a typical number is a drop of 5% per month when packed in glass and non-refrigerated. Refrigeration will prolong the potency and viability of most probiotic stability to maintain higher counts over a longer period of time.
In order to meet label claims for probiotic CFU numbers, manufacturers including NOW Foods generally add an overage to allow for the natural decline in numbers over time. We test to assure that the product meets specifications and label claims in terms of potency (CFUs) at the time of manufacture. Probiotic stability studies utilizing the strains, potencies, and designated packaging for a specific product are also done as needed to generate data to calculate an experimental stability curve that predicts changing CFU counts throughout the shelf life of the product. However, the actual rate of change depends on the environment that the product is exposed to, especially if not kept refrigerated.
The rate of decline in viable CFU numbers can increase if a probiotic product is held in conditions that are very warm or moist, especially after opening when moisture can more easily get into the package. Because of the extreme variability of the seasonal weather and environmental conditions that a product may experience, and duration of these exposures, it’s not possible to precisely predict potency and shelf life accurately for every person’s situation across a wide geographical area. In some cases, it could happen that adverse conditions may lower probiotic counts below label claim.
Refrigeration is ideal for storing and maintaining probiotic stability. But if that is not possible, it is best to keep the package in a cool, dry place to maintain good stability. Unfortunately, many people tend to keep their supplements in the kitchen or bathroom, which are notorious for being warm and moist and known to shorten the shelf life of many kinds of supplements. Those conditions are ideal for bacterial and mold growth; which means that the probiotics will first tend to activate but then die off more quickly than expected since they are not yet in the human body where they have a chance to live, grow, and thrive. In these adverse conditions, probiotics may lose potency more rapidly than anticipated and thus may not meet label claims for CFU counts that are calculated based on a cool, dry place. On the other hand, refrigerating probiotic products will enhance viability and shelf life.
In testing designed to mimic exposures during transportation in a hot climate, a sealed probiotic formula exposed to a temperature of over 122° F for 24 hours still met label claim for CFU content. This indicates that this level of heat over that time period was not enough to kill many of the organisms in the package, so it would take even harsher conditions to rapidly degrade the potency of this specific probiotic product.
There is some evidence that even non-viable probiotic organisms left in the package after being exposed to unfavorable conditions may have some utility in gut health. For example, it is hypothesized that they may take up ecological space on the intestinal walls, which may help prevent the growth of opportunistic organisms vying for that same space. For example, inactivated Lactobacillus planetarium has been shown in clinical studies to support our natural immunity to foreign substances (i.e., so-called acquired immunity).2
The stability of a probiotic formula tested at the time of manufacture will depend on a combination of factors. Variations in packaging, temperature, and humidity will affect the viability of probiotic products before they are taken. Protective factors that help to preserve the freshness and viability of the probiotic strains in a supplement include refrigeration, resistant packaging, and storage in a cool, dry place.
1 Oral Probiotics: An Introduction. NCCAM Publication No. D345, Created January 2007, Updated November 2011. The National Center for Complementary and Alternative Medicine (NCCAM), National Institutes of Health.
2 Hirose Y, Murosaki S, Yamamoto Y, Yoshikai Y, Tsuru T. Daily intake of heat-killed Lactobacillus plantarum L-137 augments acquired immunity in healthy adults. J Nutr. 2006 Dec;136(12):3069-73. PubMed PMID: 17116721.




New! Comments
Have your say about what you just read! Leave me a comment in the box below.